As dementia researchers we can be a bit brain focused. For obvious reasons, that’s the bit of the body that’s currently not functioning. But today we’re going to think of dementia and brain health in the context of the thing that carries your brain around, the rest of your body.
In making notes for this blog I scribbled down a few subtitles and the first one was ‘chicken and egg problem’. I then went back and read a previous post and realise I’ve used that phrase before. So for this post let’s describe the issue of inflammation and neurodegeneration as a Catch-22 instead. Which I’ve also used before but we’re going to have to just suck it up here people, there are only so many words and I’m not that verbose. Neurodegeneration can cause inflammation, but inflammation might cause neurodegeneration. A perfect circle of misery.
But let’s start, as always, with some basics on inflammation and what it’s for.
The immune system is made up of two main parts; the innate and the adaptive. For the purposes of this article we’ll stick with the innate immune system but we might talk about T-cells at a later date. The role of the innate immune system is to act as the first line of defence against the worst scum of the universe (a Men in Black reference, for those of you old enough to remember it). Let’s say you scrape your hand falling off your bike. The pavement isn’t the cleanest place and so you’re going to want to prevent any bugs from taking serious hold in your blood stream and giving you sepsis. So what happens is local cells in your skin put out the signal that damage has happened. This signal recruits cells that normally float around in your circulation, waiting to be called on, and they rock up and start causing chaos. They consume any bacteria hanging around, but they also fling out a signal for more cells to come and join the fight. The fight involves chucking out things like reactive oxygen species and reactive nitrogen species, which help kill bacteria.
Sounds useful, right? Yes and no. It is incredibly useful when you’re in tissue like the skin, where you can make more of it. But when you’re in tissue like the brain, where many of the cells are a one-shot deal, then we have more of a problem.
I know I said we’d be talking about the rest of the body but here we need to start inside the brain. Bear with me. Within the brain itself there are resident immune cells; microglia. The purpose of these cells is to keep an eye on the brain microenvironment, they’re essentially tissue macrophages in your brain. They survey things and scavenge things. So when you have a traumatic brain injury, for example, they help clear up all the dead tissue which you do not want hanging around in your brain.
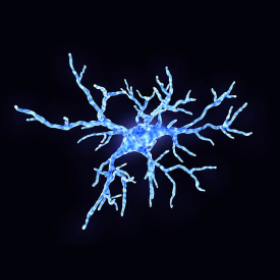
Microglia cells are the immune cells of the central nervous system and they play an important role in brain infections and inflammation. In vivo imaging studies have revealed that in the resting healthy brain, microglia are dynamic, moving constantly to actively survey the brain.
One of the first studies to notice increased numbers of reactive microglia in the brains of dementia patients was by Edith and Patrick McGeer in 1987. They found a correlation between areas of the brain containing increased numbers of plaques, and areas containing increased numbers of reactive microglia. However, as we’ve already established correlation does not equal causation. Pirates do not cause hurricanes. And in the 30 years since we’ve been trying to establish the role of microglia in the pathology of dementia.
There have been some excellent imaging studies in humans, using tracers and techniques that can detect both amyloid deposition (thought to be an important causal factor of Alzheimer’s) as well as activated microglia. These studies have demonstrated that you often get microglial activation in cognitively impaired individuals in the absence of amyloid pathology. This suggests, potentially, that inflammation within the brain that might precipitate dementia. And there are various other lines of evidence which also suggest this. Big genome wide association studies have found that genes associated with microglial activation are more frequently mutated in Alzheimer’s. We also know that mild traumatic brain injuries, such as those which might be experienced by rugby players or boxers, result in increased neuroinflammation and are associated with an increased risk of dementia.
This inflammation within the CNS may result in a positive-feedback loop whereby inflammatory molecules precipitate amyloid pathology which, in turn, results in further inflammation. But could this inflammatory pathology within the brain all start elsewhere? To understand that we need to know a little about how the brain and the body, specifically the systemic immune system, communicate with each other.
Now…this is my field of fun. I love the fact that this communication axis is vital for so many diseases and yet so much of it remains unmapped. So I’ll try and cover some of the basics about what we do know.
The brain used to be called ‘immune privileged’ because it was thought it wasn’t subject to the same immune surveillance by circulating cells as the rest of the body. It has the blood brain barrier, a network of vessels containing specialised tight junctions which, in theory, keep most circulating cells out. However, there are bits of the brain where this barrier is less…present, for want of a better word. The circumventricular organs, for example. These are particular areas of the brain, such as the hypothalamus, where large molecules like hormones need to access the brain. And these areas can also be where things like circulating inflammatory mediators like cytokines get in. Various other mechanisms, including vagal nerve stimulation and transcytosis across the blood brain barrier have been proposed as mechanisms for how systemic inflammation might cause inflammation within the CNS.
And we’ve just established up top that inflammation within the CNS might precipitate the pathology of dementia. So in a spider-ate-the-fly kind of way, might systemic inflammation cause central inflammation which then causes dementia?
There is some evidence this might be the case but it’s very broad population-based stuff. There were a number of big epidemiological studies in the ‘90s which showed that patients on NSAIDs (non-steroidal anti-inflammatory drugs – things like ibuprofen) for other diseases, had a lower risk of developing dementia. But studies looking at anti-inflammatory drugs in clinical trials have not found them to be effective. This, in and of itself, is interesting. It’s possible that what various authors have stumbled upon here is the precipitating factor. Inflammation might be important before the disease starts, but once it gets going the inflammation is really a side show.
And that side show involves some of those basic cells I talked about at the start getting out of your blood stream and into your brain. Things like neutrophils, which are very effective killers of bacteria, migrate into your brain parenchyma and – because they’re activated – start chucking out things like reactive oxygen species. Neurons, always a delicate bunch to begin with, don’t cope well with this change in their finely tuned microenvironment and promptly start to get stressed and die. There have been some very elegant studies in mice to show that if you inhibit neutrophil migration in transgenic models of Alzheimer’s, you can delay cognitive impairment.
There are also inflammatory pathologies which are associated with increased risk of developing dementia. Things like rheumatoid arthritis or inflammatory bowel disease. The latter is particularly interesting because the gut microbiome produces its own amyloids, and there has been suggestion that these are capable of ‘seeding’ the CNS. LPS, lipopolysaccharide, is molecule found on the outer membrane of bacteria like E.Coli, which are prevalent in your gut. If you inject this into mice, you find that amyloid pathology in the brain starts earlier than it would under normal circumstances.
And there’s been evidence to demonstrate that if you have increased levels of inflammation around mid-life, which persist, then you’re much more likely to develop dementia, or have cognitive issues. And because the brain and the immune system are so intrinsically linked there are all sorts of things which might cause inflammation. Like stress. So I better quit frenetically writing in case I set off my immune system and precipitate my own decline into dementia.

Dr Yvonne Couch
Author
Dr Yvonne Couch is an Alzheimer’s Research UK Fellow at the University of Oxford. Yvonne studies the role of extracellular vesicles and their role in changing the function of the vasculature after stroke, aiming to discover why the prevalence of dementia after stroke is three times higher than the average. It is her passion for problem solving and love of science that drives her, in advancing our knowledge of disease. Yvonne has joined the team of staff bloggers at Dementia Researcher, and will be writing about her work and life as she takes a new road into independent research.

 Print This Post
Print This Post




